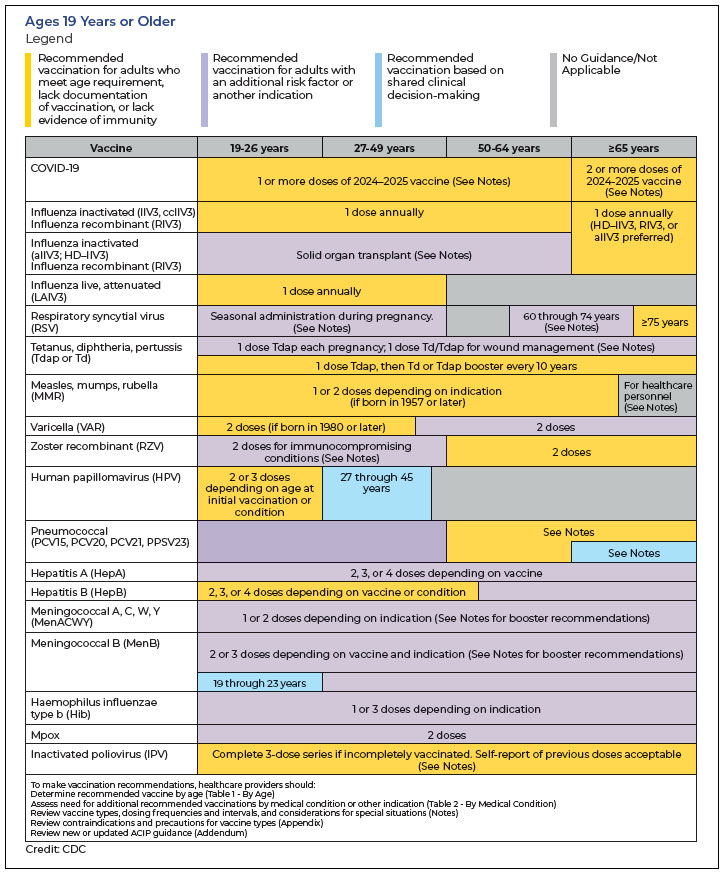
On October 29, 2024, the Centers for Disease Control and Prevention (CDC)’s Advisory Committee on Immunization Practices (ACIP) made some recommendations that are going to change the way immunization schedules look for 2025, particularly as they relate to pneumococcal disease and COVID-19.1
The most significant changes are that the CDC lowered the age for routine adult pneumococcal vaccination, from 65 to 50 years old, and that the CDC now recommends a second dose of the 2024-2025 COVID-19 vaccine for people aged ≥56 years who are moderately or severely immunocompromised 6 months after their most recent 2024-2025 COVID-19 vaccine dose.
Just 2 weeks after these recommended changes, a speaker at the Joint Federal Pharmacy Seminar (JFPS) gave a timely talk on the updated immunization recommendations, specifically regarding pneumococcal disease prevention in adults.
In a presentation theater at the meeting that was supported by Merck, Jeffrey Goad, PharmD, MPH, president of the National Foundation for Infectious Diseases (NFID), reviewed the ongoing burden and shifting epidemiology of pneumococcal disease in adults, and discussed the ACIP’s changes.2
Despite the potential severity of pneumococcal disease, a recent NFID national survey concluded that among those at higher risk for pneumococcal disease, only 44% planned to get vaccinated; the top reason cited for avoiding vaccination was concern about potential vaccine-related adverse events.3
Dr Goad told those in attendance at JFPS that pharmacists are in a unique position to educate the public about their vaccination options, and emphasized the importance of vaccination in preventing illness.
Pneumococcal pneumonia is a leading bacterial cause of hospitalized pneumonia in the United States. Older adults and people with lung disease and other chronic medical conditions or risk factors are at increased risk of serious disease or death from pneumococcal pneumonia.
“The new recommendation gives more US adults the opportunity to help protect themselves from pneumococcal disease, which can lead to serious illnesses, including pneumonia, meningitis, and bloodstream infections,” NFID Medical Director Robert H. Hopkins, Jr, MD, said in a related NFID press release.3 “Vaccination is our best defense against these serious diseases. We want everyone who is eligible to take advantage of this opportunity to help lower their risk.”
There are 3 pneumococcal conjugate vaccines (PCV15, PCV20, PCV21) available for adult vaccination, according to the CDC’s website. ACIP officials did not state a preference for any particular vaccine, but on their website, CDC officials note, “typically, only 1 dose of pneumococcal vaccine is needed as an adult. However, if PCV15 is used, a second type of vaccine (pneumococcal polysaccharide vaccine, PPSV23) is needed.”1
Pneumococcal vaccines are recommended for children aged <5 years and adults aged ≥50 years, as well as for children and adults at increased risk for pneumococcal disease. These vaccines can be given at the same time as other recommended vaccines, including:
- Respiratory syncytial virus vaccines (recommended for pregnant women, adults aged ≥75 years, and adults aged 60 to 74 years who have certain risk factors like asthma and chronic obstructive pulmonary disease)
- Annual flu vaccines and updated COVID-19 vaccines (recommended for everyone aged ≥6 months). At the same 2024 meeting where the CDC announced the change in recommended ages, they endorsed a recommendation for people aged ≥65 years and for immunocompromised individuals to receive a second dose of the 2024-2025 COVID-19 vaccine
Regarding COVID-19, CDC officials wrote on their website: “Data continue to confirm the importance of vaccination to protect those most at-risk for severe outcomes of COVID-19. Also, data on COVID-19 vaccine effectiveness indicate that protection against COVID-19–associated emergency department and urgent care visits and hospitalization likely decreases by 4 to 6 months after vaccination. Fortunately, protection from admission to the intensive care unit, a sign of critical illness, lasts longer.
In addition, data show that SARS-CoV-2, the virus that causes COVID-19, continues to circulate year-round, with peaks typically occurring in the winter and late summer.
The CDC previously recommended that people who are moderately or severely immunocompromised get additional doses of the COVID-19 vaccine, in consultation with a healthcare provider. Historically, uptake of these additional doses for this group has been low. ACIP voted to provide a clear recommendation for people aged ≥6 months and who are moderately or severely immunocompromised to receive a second dose of the 2024-2025 COVID-19 vaccine 6 months after their first dose.
These updated recommendations also allow for flexibility for additional doses (ie, 3 or more) for people who are moderately or severely immunocompromised, in consultation with their healthcare provider (a strategy known as shared clinical decision-making). These additional doses can be timed around immunosuppressive treatments such as chemotherapy, after which some people may be at increased risk of severe COVID-19.”
References
- Centers for Disease Control and Prevention. CDC updates vaccine recommendations. Published October 29, 2024. Accessed December 9, 2024. www.cdc.gov/ncird/whats-new/cdc-updates-vaccine-recommendations.html
- Goad J. Presentation theater: recognizing and addressing the risks of shifting epidemiology of adult pneumococcal diseases. Presented at: Joint Federal Pharmacy Seminar. November 9-12, 2024; San Diego, CA.
- National Foundation for Infectious Diseases (NFID). Adults age 50 and older can help lower risk for pneumonia. Press release. Published November 8, 2024. Accessed November 13, 2024. https://www.nfid.org/adults-age-50-and-older-can-help-lower-risk-for-pneumonia/