Introduction
The natural histories and outcomes of upper gastrointestinal (GI) tract cancers differ based on the location of disease and are subdivided into esophageal, gastric, and gastroesophageal junction cancers (GEJC).1,2 Esophageal cancer (EC) is estimated to account for 22,370 cases and 16,130 deaths in the United States in 2025, representing 1.1% and 2.6% of all cancers and cancer deaths, respectively.3 Gastric cancer (GC), which shares similar epidemiology, is estimated to account for 30,300 cases and 10,780 deaths in the United States in 2025.4 For both EC and GC, patients are most frequently diagnosed between ages 65 to 74 years, with the median age at diagnosis being 68 and 69 years, respectively.3,4 Tobacco use is an associated risk factor for both EC and GC.1,5 Risk factors for EC also include heavy alcohol use and Barrett’s esophagus.1 Other risk factors for GC are gastritis, obesity, gastroesophageal reflux disease, and genetics.5
Esophageal squamous cell carcinoma (ESCC) and adenocarcinoma are the most common types of EC, with ESCC dominating the upper and middle esophagus and adenocarcinoma in the lower esophagus.1 GC and GEJC are most frequently adenocarcinoma.2,6 Most EC and GC/GEJC cases are metastatic at diagnosis, and survival outcomes in both are poor. From 2014 to 2020, 18% of EC cases were localized at diagnosis with a 5-year survival rate of 48.1%. During that same time, a greater number of EC cases were metastatic at diagnosis (39%) with a comparatively greatly reduced 5-year survival rate (5.3%). GC follows a similar trend, with 29% and 36% of GC cases diagnosed at the localized and metastatic stage and 5-year survival outcomes at 75.4% and 7.0%, respectively.4 GEJC presents and recurs more aggressively compared with GC, leading to poorer clinical outcomes.7
The current treatment paradigms of advanced, unresectable or metastatic ESCC and GC/GEJC utilize a fluoropyrimidine and a platinum agent as the chemotherapy backbone. Per NCCN guidelines, first-line (1L) systemic therapy of advanced ESCC and GC/GEJC primarily consists of combination therapy with a fluoropyrimidine and a platinum agent, while second-line (2L) treatment may consist of a single-agent taxane or irinotecan.8,9 Anti–programmed cell death protein 1 (PD-1) antibodies are also recommended by NCCN in both 1L and 2L treatment of advanced ESCC and GC/GEJC, whether in combination with chemotherapy (1L) or as a single agent (2L).8,9 However, the antitumor response of anti–PD-1 antibodies may not be as robust as desired. Particularly, anti–PD-1 antibodies that bind to Fc gamma receptors (FcγRs) likely mediate the cross-linking between PD-1–positive T cells and FcγR-positive macrophages, increasing T-cell clearance and possibly diminishing antitumor responses.10
Novel, improved treatments are needed. Tislelizumab-jsgr recently gained expanded use within GCs and is currently approved for 3 indications11:
- In combination with platinum-containing chemotherapy for 1L treatment of adults with unresectable or metastatic ESCC whose tumors express programmed cell death-ligand 1 (PD-L1) (≥1)
- As a single agent in adults with unresectable or metastatic ESCC after prior systemic chemotherapy that did not include a PD-L1 inhibitor
- In combination with platinum and fluoropyrimidine-based chemotherapy in adults for the 1L treatment of unresectable or metastatic HER2-negative GC/GEJ adenocarcinoma whose tumors express PD-L1 (≥1)
This article offers an overview of tislelizumab, recent clinical data, and current approved indications in ESCC and GC/GEJC, and defines optimal administration of treatment.
Commentary by David Wang, MD, PhD: There are 2 major histologic types of esophageal cancer, esophageal squamous cell carcinoma, which is highly prevalent worldwide, and esophageal adenocarcinoma, which is more prevalent in the Western world. In terms of etiology and treatment, esophageal adenocarcinoma is typically grouped with gastroesophageal junction cancers and gastric cancers. The therapeutic advancement in these diseases has been the addition of immunotherapy to chemotherapy. In upper GI cancers, if patients are PD-L1–positive, treatment is typically some type of chemotherapy doublet, which most commonly includes fluoro-pyrimidines, with immunotherapy.
Overview of Tislelizumab-jsgr
Tislelizumab is a humanized IgG4 monoclonal antibody that blocks the interaction between PD-1 and PD-L1/2. This interaction reduces PD-1 pathway–mediated suppression of the immune response, resulting in a greater antitumor response.11 While tislelizumab is one of many PD-1 inhibitors on the market, tislelizumab differentiates from currently marketed assets such as pembrolizumab and nivolumab in several important ways.
Firstly, tislelizumab has minimal binding to FcγR on macrophages compared with most anti–PD-1 therapies, leading to minimal T-cell clearance and an enhanced antitumor response.10 Furthermore, tislelizumab has a unique binding orientation and kinetics toward PD-1 compared with other PD-1 inhibitors. This is complemented by a high target affinity and slow dissociation rate from PD-1 that results in a >99% blockade of the PD-1/PD-L1 interaction and a similar half-life to that of pembrolizumab and nivolumab, respectively.10-13
Commentary by David Wang, MD, PhD: Tislelizumab is more selective, binds more effectively, [and] enhances T-cell activity. I think for healthcare professionals, it is important that the dissociation constant is much higher for tislelizumab than other PD-1 inhibitors. Furthermore, it is important that tislelizumab has epitopes it’s targeting that pembrolizumab and nivolumab don’t, by design. If an anti–PD-1 antibody binds PD-1 on a T cell, you’re blocking interaction with PD-L1 and PD-L2, enhancing T-cell activity, but if the Fc portion of the antibody binds the FcγR on a macrophage, the macrophage can then phagocytize that T cell. Since tislelizumab doesn’t bind [to the] FcγR on macrophages, it makes sense intellectually to healthcare professionals that tislelizumab would induce an augmented immune response.
Clinical Data: Phase 3 RATIONALE-306
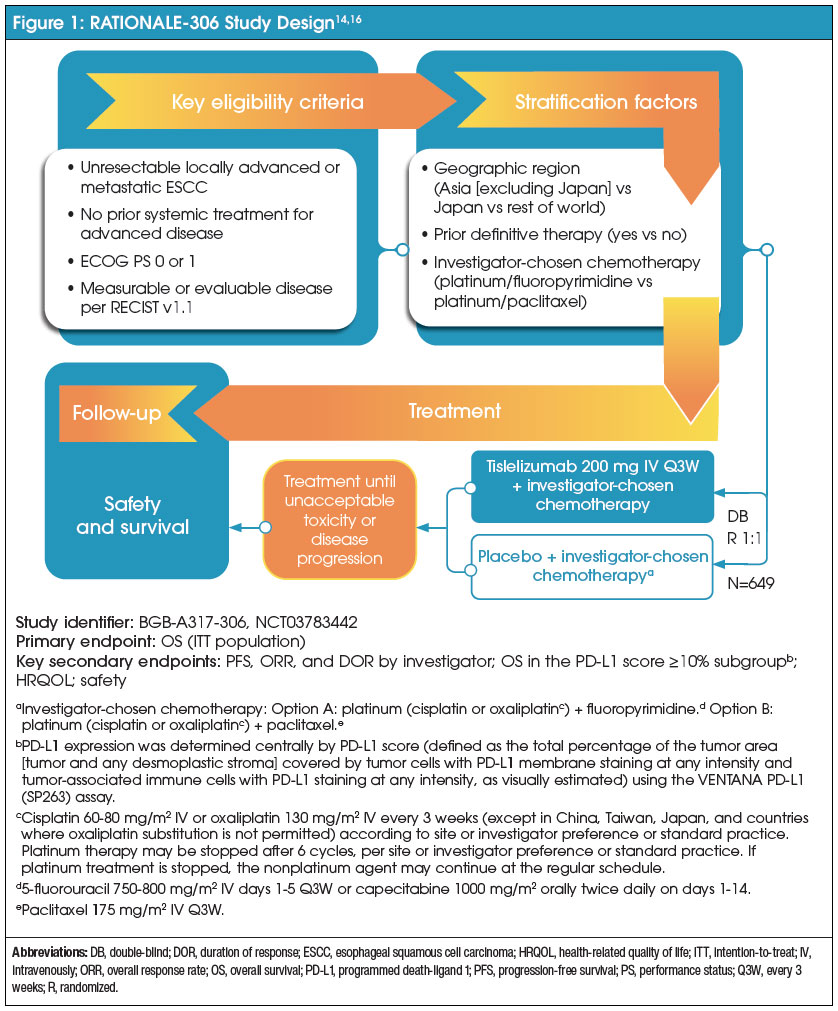
RATIONALE-306 (NCT03783442) is a phase 3 study of tislelizumab in combination with an investigator-chosen chemotherapy versus investigator-chosen chemotherapy alone in adult patients with unresectable, locally advanced, recurrent, or metastatic ESCC.14 The trial enrolled 649 patients regardless of PD-L1 expression with an ECOG performance status of 0 to 1 and measurable disease per RECIST v1.1.14 Figure 1 demonstrates the study design visually. Although patients were enrolled regardless of PD-L1 expression level, PD-L1 expression levels were evaluated via tumor area positivity (TAP) at a central laboratory utilizing the VENTANA PD-L1 (SP263) assay.11 TAP is a method of PD-L1 expression evaluation similar to combined positive score (CPS) and is calculated with the percentage of PD-L1–positive cells divided by the tumor area. It has been shown that TAP scoring requires reduced time compared with CPS while demonstrating a high concordance rate with CPS.15
Patients were randomized 1:1 to receive tislelizumab 200 mg (n=326) or placebo (n=323) intravenously every 3 weeks on day 1 of their cycle in combination with an investigator-chosen chemotherapy doublet.14 An investigator-chosen chemotherapy consisted of a platinum agent (cisplatin 60-80 mg/m2 intravenously on day 1 or oxaliplatin 130 mg/m2 intravenously on day 1) plus a fluoropyrimidine (fluorouracil 750-800 mg/m2 intravenously on days 1-5 or capecitabine 1000 mg/m2 orally twice daily on days 1-14) or paclitaxel 175 mg/m2 intravenously on day 1.14 When a patient receives paclitaxel, cisplatin and oxaliplatin may be given on day 1 or 2.11
Baseline patient and disease characteristics between the 2 treatment groups were similar. Approximately 86% and 14% of patients showed metastatic and locally advanced disease, respectively.11,14 Thirty-four percent of patients had tumors that expressed PD-L1 TAP ≥10%, and 74% had PD-L1 TAP <1%.11
The primary endpoint was overall survival (OS) for the intention-to-treat (ITT) population, defined as the time from the date of randomization until the date of death due to any cause.14,16 Key secondary endpoints included progression-free survival (PFS), objective response rate (ORR), duration of response (DOR), and OS in the PD-L1 ≥10% subgroup.14 Safety was also evaluated in this trial.
Efficacy
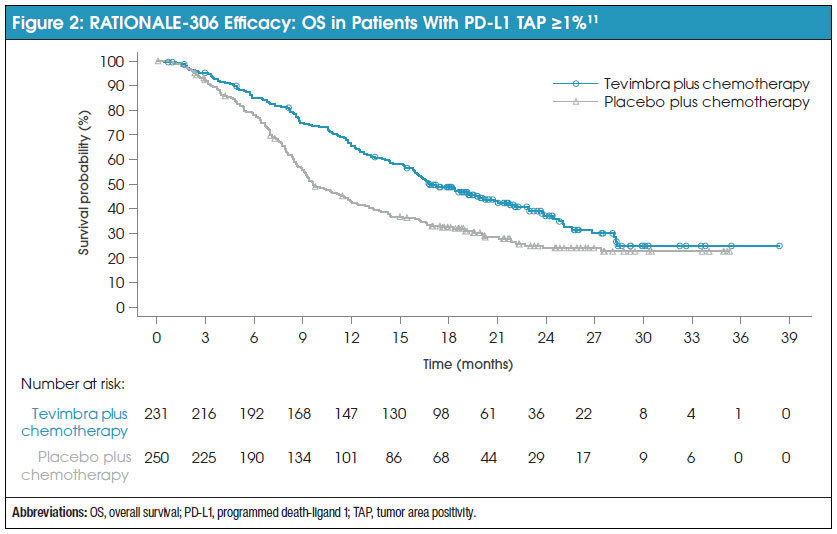
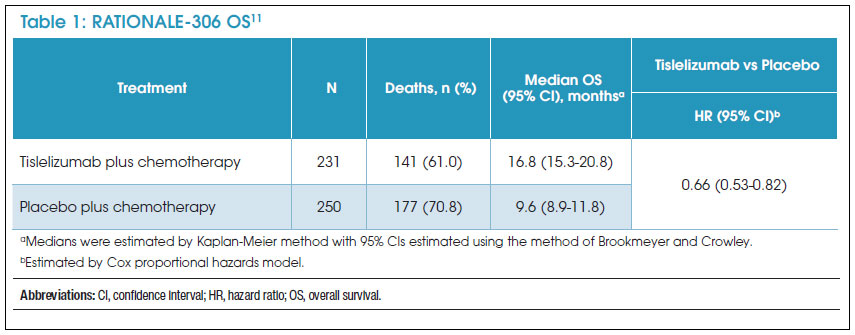
After a median follow-up of 16.3 months (interquartile range, 8.6-21.8), median OS for patients with PD-L1 TAP ≥1% in the tislelizumab group was 16.8 months (95% confidence interval [CI], 15.3-20.8 months) compared with 9.6 months (95% CI, 8.9-11.8 months) in the placebo group.11,14 These results were statistically significant and demonstrated a greater survival benefit of tislelizumab in combination with chemotherapy compared with chemotherapy alone (hazard ratio [HR], 0.66 [95% CI, 0.53-0.82]).11 Figure 2 and Table 1 show the Kaplan-Meier survival curve and survival data for patients with a PD-L1 TAP score of ≥1%. Secondary endpoints also supported tislelizumab.11 A progression benefit was seen in the tislelizumab arm, with 152 PFS events (66%) in the tislelizumab group compared with 199 (80%) in the placebo group (HR, 0.56 [95% CI, 0.45-0.70]).11 ORR (58% vs 36%) and DOR (7.2 months [95% CI, 6.2-9.6] vs 5.7 months [95% CI, 4.4-7.3]) also showed improvement for patients in the tislelizumab group.11 While TAP was utilized as the method of PD-L1 expression measurement, a retrospective analysis of the efficacy data determined that the utilization of CPS scores yielded generally consistent results within TAP subgroups.11
Commentary by David Wang, MD, PhD: RATIONALE-306 showed an overall survival benefit for tislelizumab for the first-line treatment of metastatic ESCC in combination with chemotherapy. With no new safety events, I think it’s confirmatory as the third PD-1 inhibitor that’s active in this disease. If you had to pick 1 thing that differentiated this trial with tislelizumab, it’s that RATIONALE-306 used oxaliplatin and paclitaxel. This data gives healthcare providers the flexibility to choose a different chemotherapy backbone in the treatment of metastatic ESCC, especially chemotherapy regimens that are preferred by American oncologists.
Safety
Treatment-emergent adverse events (TEAEs) occurred in 313 (97%) and 309 (96%) patients in the tislelizumab and placebo groups, respectively.14 The most common grade ≥3 TEAEs for the tislelizumab versus placebo groups were decreased neutrophil count (31% vs 33%), decreased white blood cell count (11% vs 16%), and anemia (15% vs 13%), respectively.14
These findings may help to highlight the adverse-event (AE) profile of tislelizumab and assist healthcare providers in preparing their center to utilize this agent. However, the safety and efficacy data demonstrate that tislelizumab in combination with chemotherapy is superior to chemotherapy alone in 1L treatment of advanced or metastatic ESCC, with an AE profile that is to be expected of the individual treatment components.
Commentary by David Wang, MD, PhD: From RATIONALE-306, tislelizumab showed a slightly lower adverse-event rate, or of at least grade 3 or higher adverse events compared with other PD-1 inhibitors. As expected, there were more immune-related adverse events with tislelizumab as compared to placebo, which is seen with other PD-1 agents too.
Clinical Data: Phase 3 RATIONALE-302
RATIONALE-302 (NCT03430843) was the basis of the initial ESCC indication for tislelizumab, and was a global, open-label, phase 3, randomized controlled trial that evaluated patients who progressed during or after 1L therapy for advanced ESCC.17 Patients were recruited into the study regardless of PD-L1 expression and were randomized 1:1 to receive either tislelizumab 200 mg (n=256) intravenously every 3 weeks or an investigator-chosen chemotherapy (n=256).17 An investigator-chosen chemotherapy consisted of single-agent paclitaxel, docetaxel, or irinotecan.17 Like RATIONALE-306, PD-L1 expression level was evaluated via TAP at a central laboratory.11
Baseline characteristics of the tislelizumab and placebo groups were similar.17 Approximately 84% of patients were male, and patients in the tislelizumab and placebo groups had a median age of 62 years.11 Most patients were of Asian ethnicity.17 PD-L1 expression level was <10% for 43% of patients.11 The primary endpoint of the study was OS in the ITT population, and secondary endpoints included OS in patients with PD-L1 TAP ≥10%, PFS, ORR, and DOR.17
Efficacy
After a median follow-up time of 8.5 and 5.8 months, the median OS in all patients was 8.6 months (95% CI, 7.5-10.4) and 6.3 months (95% CI, 5.3-7.0) for the tislelizumab and placebo groups, respectively.11,17 The benefit was maintained for patients with PD-L1 TAP ≥10%, demonstrating a median OS of 10.3 months (95% CI, 8.5-16.1) and 6.8 months (95% CI, 4.1-8.3) in the tislelizumab and placebo groups, respectively (HR, 0.54 [95% CI, 0.36-0.79]).17 Secondary endpoints also favored tislelizumab. Median PFS (1.6 months vs 2.1 months), ORR (15.2% vs 6.6%), and DOR (10.3 months vs 6.3 months) were all greater in the tislelizumab group compared with placebo.11 Unlike in RATIONALE-306, concordance of efficacy results when utilizing CPS or TAP scoring to evaluate PD-L1 expression was not evaluated in RATIONALE-302.11
Safety
Patients being treated with tislelizumab experienced decreased rates of treatment-related AEs (TRAEs) compared with placebo (18.8% vs 55.8%).17 TRAEs led to treatment discontinuation in 16% of patients in the tislelizumab group compared with 8% in the placebo group.17 Serious AEs occurred in 41% of patients, with the most frequent AEs (≥2%) being pneumonia, dysphagia, hemorrhage, pneumonitis (including pneumonitis and immune-mediated pneumonitis), and esophageal obstruction.11 The most common grade 3 or 4 AEs for both the tislelizumab and placebo groups were similar, including pneumonia (6% vs 7%), anemia (6% vs 11%), dysphagia (6% vs 3%), fatigue (2% vs 6%), abdominal pain (0.8% vs 2%), and vomiting (0.8% vs 4%), respectively.11
As was seen in RATIONALE-306, tislelizumab provided a statistically significant and clinically meaningful improvement in OS compared with chemotherapy alone in patients with advanced or metastatic ESCC who had disease progression after 1L systemic therapy. Tislelizumab also demonstrated a tolerable safety profile, with lower rates of most grade ≥3 AEs.
Clinical Data: Phase 3 RATIONALE-305
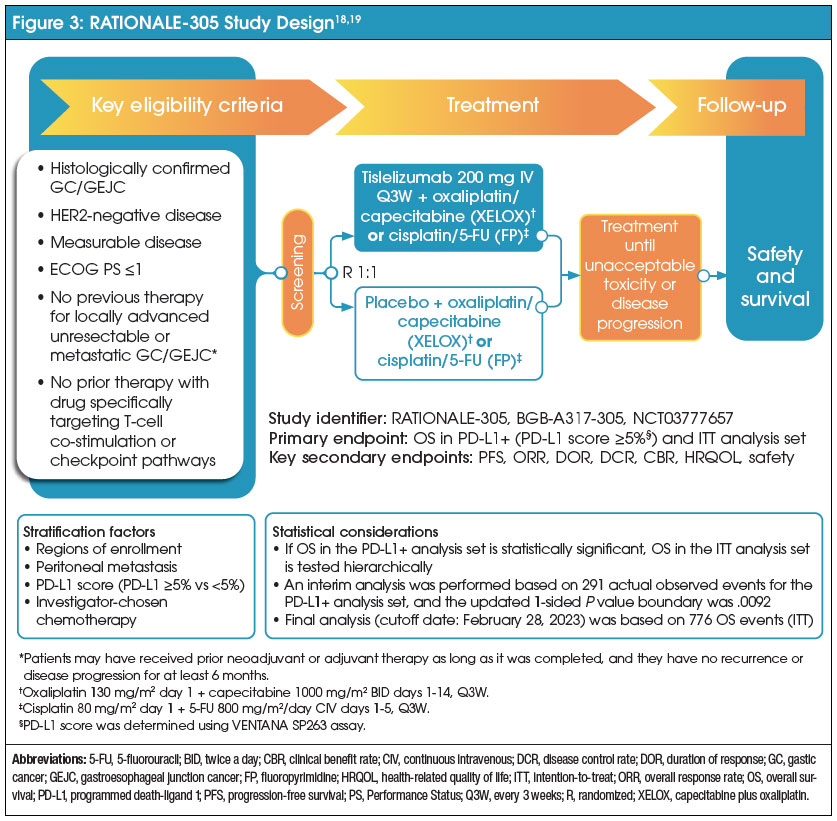
Tislelizumab has also been recently approved in the treatment of GC/GEJ adenocarcinoma. RATIONALE-305 (NCT03777657) was a phase 3, double-blind, placebo-controlled trial that enrolled 1657 adult patients with HER-2 negative, locally advanced, unresectable or metastatic GC/GEJ adenocarcinoma, regardless of PD-L1 expression status.18 Patients who received prior systemic anticancer therapy for advanced disease were excluded. This study randomized patients 1:1 to receive either tislelizumab 200 mg (n=501) or placebo (n=496) every 3 weeks in combination with an investigator-chosen chemotherapy, which consisted of capecitabine 1000 mg/m2 twice daily on days 1 to 14 and oxaliplatin 130 mg/m2 on day 1, or 5-fluorouracil 800 mg/m2 on days 1 to 5 and cisplatin 80 mg/m2 on day 1 for up to 6 cycles.18 Figure 3 provides a visual depiction of the study design.
Baseline characteristics were similar between the 2 groups.18 Patients enrolled in the trial were a median age of 61 years.11 Approximately 69% of patients were male.11 Seventy-five percent of patients were of Asian ethnicity, and approximately 80% had the stomach as the primary location of their tumor.11 In addition, 89% of patients had a PD-L1 TAP score of ≥1%.11 The primary efficacy endpoint of the study was OS in patients with a PD-L1 TAP score of ≥5% and in all randomized patients.18 Secondary efficacy endpoints were PFS, ORR, and DOR.18
Efficacy
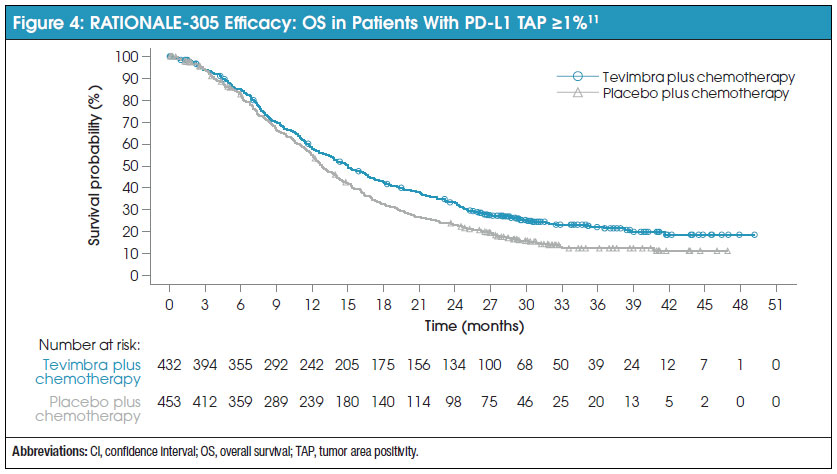
In all randomized patients, after a median follow-up of 13.2 months, tislelizumab in combination with chemotherapy demonstrated a statistically significant survival benefit compared with placebo (HR, 0.78 [95% CI, 0.67-0.90]), with a median OS benefit of 15.0 (95% CI, 13.3-16.7) and 12.8 (95% CI, 12.1-14.1) months, respectively.11 Tislelizumab’s survival benefit was conserved in patients with a PD-L1 TAP score of ≥5% (HR, 0.74 [95% CI, 0.59-0.94]).18 Figure 4 shows the Kaplan-Meier survival curves for patients with a PD-L1 TAP score of ≥1%. Secondary endpoints including PFS (HR, 0.78 [95% CI, 0.67-0.91]), ORR (tislelizumab vs placebo: 48% vs 41%), and DOR (tislelizumab vs placebo: 8.6 months vs 7.2 months) all showed improvement for the tislelizumab group.11
In addition to the prespecified analysis, a retrospective analysis on the patient population was conducted to determine the concordance of efficacy data when evaluating patient PD-L1 expression utilizing either TAP or CPS. OS between both groups was similar whether evaluating PD-L1 expression by TAP (HR, 0.78 [95% CI, 0.67-0.90]) or CPS (HR, 0.78 [95% CI, 0.67-0.91]).11 This trend was conserved for PFS and ORR.11
Commentary by David Wang, MD, PhD: Researchers compared TAP versus CPS and found not much difference. The trial investigators re-scored participants utilizing CPS and found similar benefits in clinical outcomes with tislelizumab versus placebo in patients who were CPS or TAP positive. In terms of the median overall survival, the difference between the 2 scoring methods maybe differed by a few weeks, but you could definitely differentiate the treatment groups. These results basically suggest that there’s high concordance between TAP and CPS positivity.
Commentary by David Wang, MD, PhD: A post-hoc analysis of RATIONALE-305 found that patients with peritoneal or liver metastases still benefit from treatment with tislelizumab. We typically think of those patients as doing more poorly.
Safety
Overall, grade ≥3 TRAEs were present in 53.8% and 49.8% of patients in the tislelizumab and placebo arms, respectively.18 The most common grade ≥3 TRAEs in the tislelizumab and placebo groups were decreased neutrophil count (12% vs 12%), decreased platelet count (11% vs 12%), neutropenia (7% vs 7%), and anemia (5% vs 7%). TRAEs led to a similar rate of dose modification in both the tislelizumab and placebo groups (71% vs 72%), but tislelizumab did see a higher rate of discontinuation compared with placebo (16% vs 8%).18 Tislelizumab also led to an increased rate of death related to TRAEs compared with placebo (1% vs <1%).18 TRAEs leading to deaths occurred in 6 patients in the tislelizumab group (unspecified death [n=4], colitis [n=1], and sepsis [n=1]) and 2 patients in the placebo group (pneumonia [n=2]).18
Similar to RATIONALE-306 and RATIONALE-302, tislelizumab showed superior OS benefit in combination with chemotherapy compared with chemotherapy alone, with a generally tolerable safety profile. Furthermore, retrospective analysis of safety data demonstrated that whether evaluating PD-L1 expression by TAP or CPS, there is no difference in efficacy of tislelizumab in the advanced GC/GEJ adenocarcinoma population. The combination of the efficacy, safety, and retrospective data presents tislelizumab as a potential new treatment option for patients with advanced GC/GEJ adenocarcinoma regardless of the modality of PD-L1 expression testing.
Administering Tislelizumab-jsgr
As tislelizumab is administered intravenously, the following is worthy of consideration. Tislelizumab should not be administered as an intravenous push or a single bolus injection, nor should it be co-administered with any other drugs through the same infusion line.11 At initial infusion, tislelizumab is administered over 60 minutes.11 Subsequent infusions may be administered over 30 minutes, depending on patient tolerability.11
Whether a patient is being treated for ESCC or GC/GEJC as a 1L or 2L treatment, patients will receive 1 of 3 dosing regimens; 150 mg of tislelizumab every 2 weeks, 200 mg every 3 weeks, or 300 mg every 4 weeks.11 Patients being treated for 2L ESCC may utilize tislelizumab as a single agent, while 1L ESCC and GC/GEJC require a combination with platinum-containing chemotherapy or platinum plus fluoropyrimidine, respectively.11 Dose reductions for tislelizumab are not recommended.11 For grade ≥3 immune-mediated adverse reactions, withholding tislelizumab is recommended.11 Permanent discontinuation is recommended for life-threatening (grade 4) or recurrent grade 3 immune-mediated reactions that require systemic immunosuppressive treatment or when there is an inability to reduce the corticosteroid dose to ≤10 mg or less of prednisone or equivalent per day within 12 weeks of initiating steroids.11
Commentary by David Wang, MD, PhD: Contraindications for any PD-1 antibody include (1) long-standing autoimmune disease, even though it’s not an absolute contraindication, because PD-1 blockade can worsen the autoimmune condition or (2) another chronic medical condition which requires long-term steroids since steroids will dampen the immune response. Another important factor is (3) what are the prior drugs the patient responded or didn’t respond to? If a patient has received a prior immunotherapy, I think many providers use the cutoff of being at least 6 months recurrence-free before considering retreating with a PD-1 antibody. Other considerations include (4) what is the status of other biomarkers if they can be molecularly targeted? Next, (5) what are the practice characteristics? In Veterans Affairs medical centers, patients can come from far distances, which can impact the choice of what the the chemotherapy backbone utilized is, as each regimen can have different administration schedules.. Some drugs are given every 3 weeks, others are given every 2 weeks. If you use continous-infusion fluorouracil, you have to use a pump. And so those patients now have to come twice in a 2-week period.
Commentary by David Wang, MD, PhD: RATIONALE-306 was important because it allowed use of a taxane in the trial. The FDA approval for tislelizumab does not go beyond stating it may be used in combination with a platinum-containing regimen, so you could use oxaliplatin, or most people would choose carboplatin with paclitaxel. Since tislelizumab was studied in an every-3-week schedule, this would be convenient with carboplatin and paclitaxel as they share a similar dosing schedule. As this combination also doesn’t require [a] mediport, [treatment] can be given through a peripheral IV. If you’re considering convenience for a patient who’s driving a long distance and only wants to come see you once every 3 weeks, and doesn’t want a mediport, that would be a patient that you would think about using tislelizumab in.
Access to Care
To access tislelizumab, BeOne provides myBeOneSupport, a centralized patient support program to assist patients with access-related needs and to connect them with support tools.20 myBeOneSupport provides financial support services including insurance verification, product support for uninsured and underinsured patients, and copay support for commercially insured patients that may result in payments as little as $0 per month.20
Aside from financial assistance, myBeOneSupport provides day-to-day living support through oncology nurse advocates, who work to connect patients to the resources they may need, including counseling services, support groups, and transportation or lodging assistance.20
Conclusion
Treatment of ESCC and GC/GEJC is complex. Current treatment paradigms rely on platinum agents and fluoropyrimidines as the backbone of treatment, with anti–PD-1 inhibitors recently becoming a standard part of ESCC and GC/GEJC treatment. Tislelizumab represents a breakthrough in both the ESCC and GC/GEJC treatment space, circumventing the pitfalls of other anti–PD-1 inhibitors that may temper the robustness of their tumor response. In combination with chemotherapy and as a single agent, tislelizumab has shown statistically significant and clinically meaningful survival benefits in the 1L and 2L treatment of ESCC and 1L treatment of GC/GEJC with a tolerable and manageable safety profile. This data and its unique binding properties make tislelizumab an optimal choice over traditional anti–PD-L1s, marking its place in the ESCC and GC/GEJC treatment paradigm. With optimal monitoring, care, and administration, tislelizumab is sure to deliver the same optimal outcomes reported in clinical trials to clinical practice.
Commentary by David Wang, MD, PhD: Tislelizumab is an important addition to our armamentarium, especially in patients who have metastatic ESCC requiring first- or second-line treatment. I think in the first-line setting for ESCC, if you strictly adhere to [the clinical trial data], then you may say, "I can choose tislelizumab because I have the option to use oxaliplatin, which is the preferred platinum agent in the chemotherapy backbone now. Or I can add a taxane to the platinum agent and use tislelizumab." Tislelizumab provides multiple options in terms of the chemotherapy backbone with a patient-friendly dosing schedule. I think on the gastric cancer side, again, it depends on what the prescriber’s preference is. Do you prefer to use a fluorouracil pump and give FOLFOX, or do you prefer to give an injection of oxaliplatin and trust that your patient is going to be compliant and take their capecitabine pills? And in the GC/GEJC setting, you could use tislelizumab with CAPOX [capecitabine+oxaliplatin]. So I think it really depends on your preference and comfort level and what the patient’s willing to do.
References
- National Cancer Institute. Esophageal cancer treatment (PDQ) — patient version. Accessed January 10, 2025. https://www.cancer.gov/types/esophageal/patient/esophageal-treatment-pdq#Keypoint2
- National Cancer Institute. What is stomach cancer? Accessed January 10, 2025. https://www.cancer.gov/types/stomach
- National Cancer Institute. Cancer stat facts: esophageal cancer. Accessed January 8, 2025. https://seer.cancer.gov/statfacts/html/esoph.html
- National Cancer Institute. Cancer stat facts: stomach cancer. Accessed January 8, 2025. https://seer.cancer.gov/statfacts/html/stomach.html
- National Cancer Institute. Stomach cancer causes and risk factors. Accessed January 10, 2025. https://www.cancer.gov/types/stomach/causes-risk-factors
- Buas MF, Vaughan TL. Epidemiology and risk factors for gastroesophageal junction tumors: understanding the rising incidence of this disease. Semin Radiat Oncol. 2013;1:3-9.
- Nakauchi M, Vos EL, Carr RA, et al. Distinct differences in gastroesophageal junction and gastric adenocarcinoma in 2194 patients: in memory of Rebecca A. Carr, February 24, 1988-January 19, 2021. Ann Surg. 2023;4:629-636.
- National Comprehensive Cancer Network. Esophageal and esophagogastric junction cancers (version 5.2024). Accessed January 8, 2025. https://www.nccn.org/professionals/physician_gls/pdf/esophageal.pdf
- National Comprehensive Cancer Network. Gastric cancers (version 5.2024). Accessed January 8, 2025. https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf
- Zhang T, Song X, Xu L, et al. The binding of an anti-PD-1 antibody to FcγRΙ has a profound impact on its biological functions. Cancer Immunol Immunother. 2018;7:1079-1090.
- BeOne. Tevimbra (tislelizumab-jsgr) [prescribing information]. US Food and Drug Administration. Accessed May 8, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761232s012lbl.pdf
- Feng Y, Hong Y, Sun H, et al. The molecular binding mechanism of tislelizumab, an investigational anti-PD-1 antibody, is differentiated from pembrolizumab and nivolumab. Cancer Res. 2019;13(Suppl):2383.
- Hong Y, Feng Y, Sun H, et al. Tislelizumab uniquely binds to the CC′ loop of PD-1 with slow-dissociated rate and complete PD-L1 blockage. FEBS Open Bio. 2021;3:782-792.
- Xu J, Kato K, Raymond E, et al. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first-line treatment for advanced or metastatic oesophageal squamous cell carcinoma (RATIONALE-306): a global, randomised, placebo-controlled, phase 3 study [published correction appears in Lancet Oncol. 2024;3:e102]. Lancet Oncol. 2023;5:483-495.
- Liu C, Fang F, Kong Y, ElGabry EA. Tumor area positivity (TAP) score of programmed death-ligand 1 (PD-L1): a novel visual estimation method for combined tumor cell and immune cell scoring. Diagn Pathol. 2023;1:48.
- ClinicalTrials.gov. A study of tislelizumab (BGB-A317) in combination with chemotherapy as first line treatment in participants with advanced esophageal squamous cell carcinoma. Accessed January 8, 2025. https://clinicaltrials.gov/study/NCT03783442?term=NCT03783442&rank=1
- Shen L, Kato K, Kim SB, et al. Tislelizumab versus chemotherapy as second-line treatment for advanced or metastatic esophageal squamous cell carcinoma (RATIONALE-302): a randomized phase III study [published correction appears in J Clin Oncol. 2024;4:486]. J Clin Oncol. 2022;40:3065-3076.
- Qiu MZ, Oh DY, Kato K, et al. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first line treatment for advanced gastric or gastro-oesophageal junction adenocarcinoma: RATIONALE-305 randomised, double blind, phase 3 trial. BMJ. 2024;385:e078876.
- ClinicalTrials.gov. Tislelizumab in combination with chemotherapy as first-line treatment in adults with inoperable, locally advanced or metastatic gastric, or gastroesophageal junction carcinoma. Accessed June 6, 2025. https://clinicaltrials.gov/study/NCT03777657
- BeOne. myBeOne Program Services. Accessed January 9, 2024. https://www.mybeonesupport.com/tevimbra/hcp/program-services/

About David Wang, MD, PhD
Dr David Wang earned his medical degree at Vanderbilt University and a PhD in Cellular and Molecular Medicine from the Johns Hopkins University. In addition, he completed a residency in internal medicine at the Johns Hopkins Hospital, followed by advanced training through a research fellowship in cancer biology and a clinical fellowship in medical oncology. He also spent a year at the National Cancer Institute as a Howard Hughes Medical Institute-National Institutes of Health Research Scholar.
Certified by the American Board of Internal Medicine, Dr Wang joined the University of Michigan faculty in 2024.
The author of numerous research articles, book chapters, and invited lectures, Dr Wang’s clinical and research interests focus on esophageal cancer, gastric cancer, and gastroesophageal junction cancer. His research lab is focused on understanding the role of aberrant developmental pathway signaling in esophageal cancer and precursor lesions.