With commentaries by

Jackie B. Broadway-Duren, PhD, DNP, APRN, FNP-BC, FAANP
Family Nurse Practitioner
Department of Leukemia
UT MD Anderson Cancer Center
Houston, TX

Mollie E. Moran, APRN-CNP
Oncology Nurse Practitioner
Hematologic Malignancies
The Ohio State University
Columbus, OH
Introduction
Chronic lymphocytic leukemia (CLL) is the most common type of leukemia, with an incidence of 4.7 per 100,000 men and women per year.1 Small lymphocytic lymphoma (SLL) and CLL are the same disease caused by abnormal lymphocytes, with CLL affecting the blood and bone marrow, and SLL targeting the lymph nodes.2 Patients with CLL/SLL are typically treated with a covalent Bruton’s tyrosine kinase inhibitor (BTKi) or a B-cell lymphoma 2 inhibitor (BCL2i), venetoclax, in first-line or second-line therapy.3,4 Although covalent BTKis are effective, many patients eventually discontinue treatment due to disease progression or intolerance.4,5 Guidelines recommend that patients who progress while on a covalent BTKi receive therapy with a different mechanism of action such as a noncovalent BTKi or a BCL2i, since the mechanism of resistance is similar among covalent BTKis.3,6
Pirtobrutinib was granted accelerated FDA approval based on the BRUIN phase 2 study in December 2023 for adults with CLL/SLL who have undergone at least 2 prior therapies including a BTKi and a BCL2i. On December 3, 2025, pirtobrutinib was granted full approval based on the BRUIN CLL-321 phase 3 study for adults with CLL/SLL who have previously been treated with a covalent BTKi and a BCL2i. Pirtobrutinib is a selective, noncovalent (reversible) BTKi designed to address the limitations of existing covalent BTKis.7-9 This article offers a brief overview of the mechanism of action of pirtobrutinib and an analysis of clinical data of patients with CLL and SLL from the phase 3 BRUIN CLL-321 study, presented at the American Society of Hematology Annual Meeting in 2024. Considerations for clinical practice, including expert commentary from Jackie Broadway-Duren, PhD, DNP, APRN, FNP-BC, FAANP, Department of Leukemia, University of Texas MD Anderson Cancer Center, and Mollie Moran, APRN-CNP, The Ohio State University, will also be discussed.
Mechanism of action: pirtobrutinib
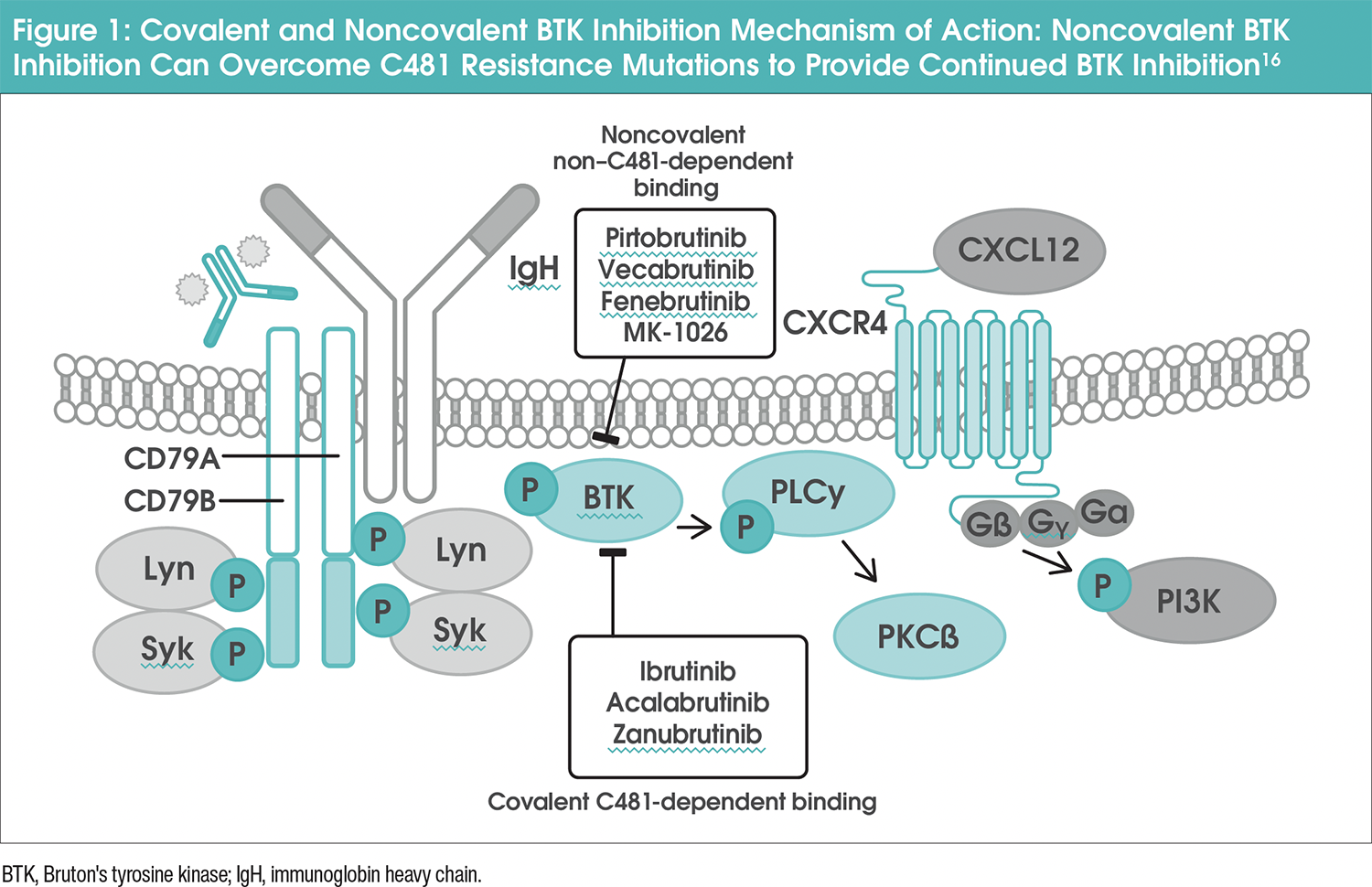
Inhibiting the Bruton’s tyrosine kinase (BTK) domain interferes with multiple pathways important for CLL cell survival, proliferation, and migration.10,11 Noncovalent inhibitors interact with BTK through reversible binding, unbinding, and rebinding, offering an alternative mechanism of inhibition.12-14 Pirtobrutinib is a highly selective, noncovalent (reversible) BTKi that can target both the wild-type (unmutated) and mutated BTK protein (Figure 1).15,16 It achieves 96% BTK target inhibition at steady-state plasma levels and has a half-life of approximately 20 hours.15 Unlike covalent BTKis, pirtobrutinib stabilizes BTK in a closed, inactive conformation, making it effective against resistant mutations and offering a promising alternative for patients with CLL or other B-cell malignancies.11,12,15,17
BRUIN CLL-321
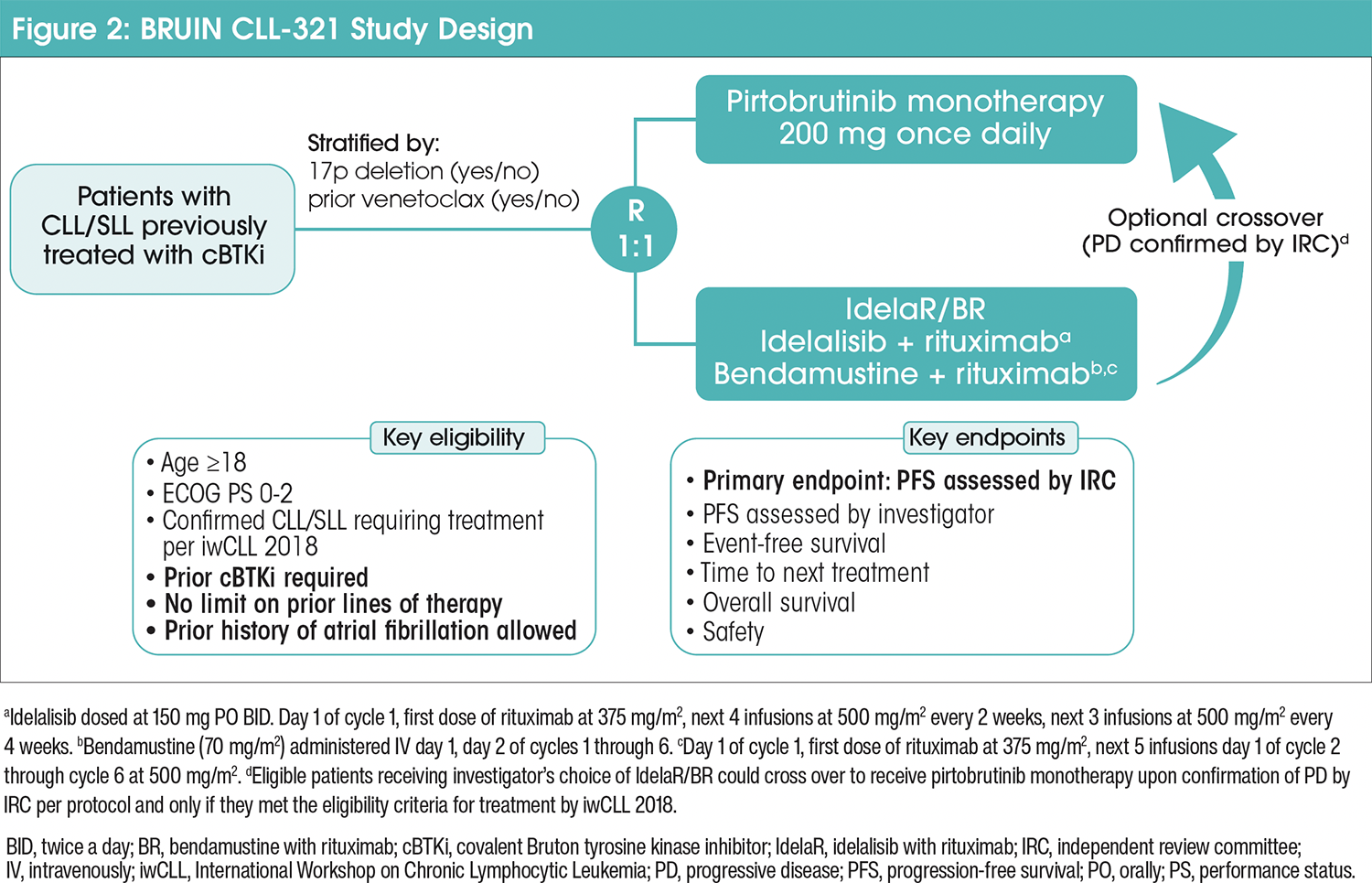
BRUIN CLL-321 was a phase 3, global, randomized, multicenter, open-label study (NCT04666038).4 This was the fi rst randomized trial conducted exclusively in patients treated with a covalent BTKi, and to date, there have been no prospective randomized controlled studies evaluating therapy for CLL/SLL after treatment with a covalent BTKi. Patients were eligible for the BRUIN CLL-321 study if they were ≥18 years of age, had an ECOG performance status (PS) of 0 to 2, and a diagnosis of CLL/SLL requiring treatment per International Workshop on Chronic Lymphocytic Leukemia (iwCLL) 2018 criteria.4 Additionally, patients must have been previously treated with a covalent BTKi, with no limit on the number of other previous therapies. Patients on concomitant anticoagulant therapy (excluding warfarin), receiving antiplatelet agents, and/or with a history of controlled atrial fi brillation at the time of enrollment were permitted.4
Eligible patients were randomly assigned 1:1 to receive either pirtobrutinib or investigator’s choice (IC) of idelalisib with rituximab (IdelaR) or bendamustine with rituximab (BR; Figure 2). Patients could cross over from IC to pirtobrutinib upon progressive disease (PD) confirmation by independent review committee (IRC), and only if they met the eligibility criteria for treatment.4
Pirtobrutinib was administered continuously at 200 mg orally once daily.4 IdelaR patients received idelalisib 150 mg orally twice a day, continuously, and rituximab 375 mg/m2 once at the first infusion, then 500 mg/m2 once every 2 weeks at the following 4 infusions, then 500 mg/m2 once every 4 weeks at the following 3 infusions. BR patients received bendamustine 70 mg/m2 intravenously, once on days 1 and 2 of up to 6 total 28-day cycles, rituximab 375 mg/m2 once at the first infusion, then 500 mg/m2 once on day 1 of cycles 2 to 6. Patients on continuous treatment remained on treatment until PD or unacceptable toxicity.4
The primary endpoint was progression-free survival (PFS) as assessed by a blinded IRC using iwCLL 2018 criteria.4 The secondary endpoint was overall survival (OS), and additional secondary endpoints included PFS by investigator, overall response rate, event-free survival, time to next treatment (TTNT) or death, and safety.4
Results of the BRUIN CLL-321 study
Between March 9, 2021, and July 17, 2023, a total of 238 patients with relapsed/refractory (R/R) CLL/SLL were randomly assigned to receive pirtobrutinib (n=119) or IC (n=119) of IdelaR (n=82) or BR (n=37).4 Baseline patient characteristics were generally balanced between groups as well as stratification factors, with 50.4% of patients in each group receiving previous venetoclax treatment. The median age was 66 years, 70% were male, and 58% had an ECOG PS ≥1. The prevalence of high-risk features was similar between pirtobrutinib and IdelaR/BR groups in patients with central results, including a TP53 mutation and/or del(17p) mutation (54% vs 54%) and a del(11q) (19% vs 25%). Some high-risk genomic features appeared more prevalent in the pirtobrutinib group versus the IdelaR/BR group, including a complex karyotype (72% vs 59%) and an unmutated immunoglobulin heavy-chain variable (IGHV) gene (93% vs 80%). The most frequent reasons for discontinuation of previous covalent BTKis in the pirtobrutinib and IdelaR/BR groups were PD (71% vs 73%) and toxicity (17% vs 18%).4
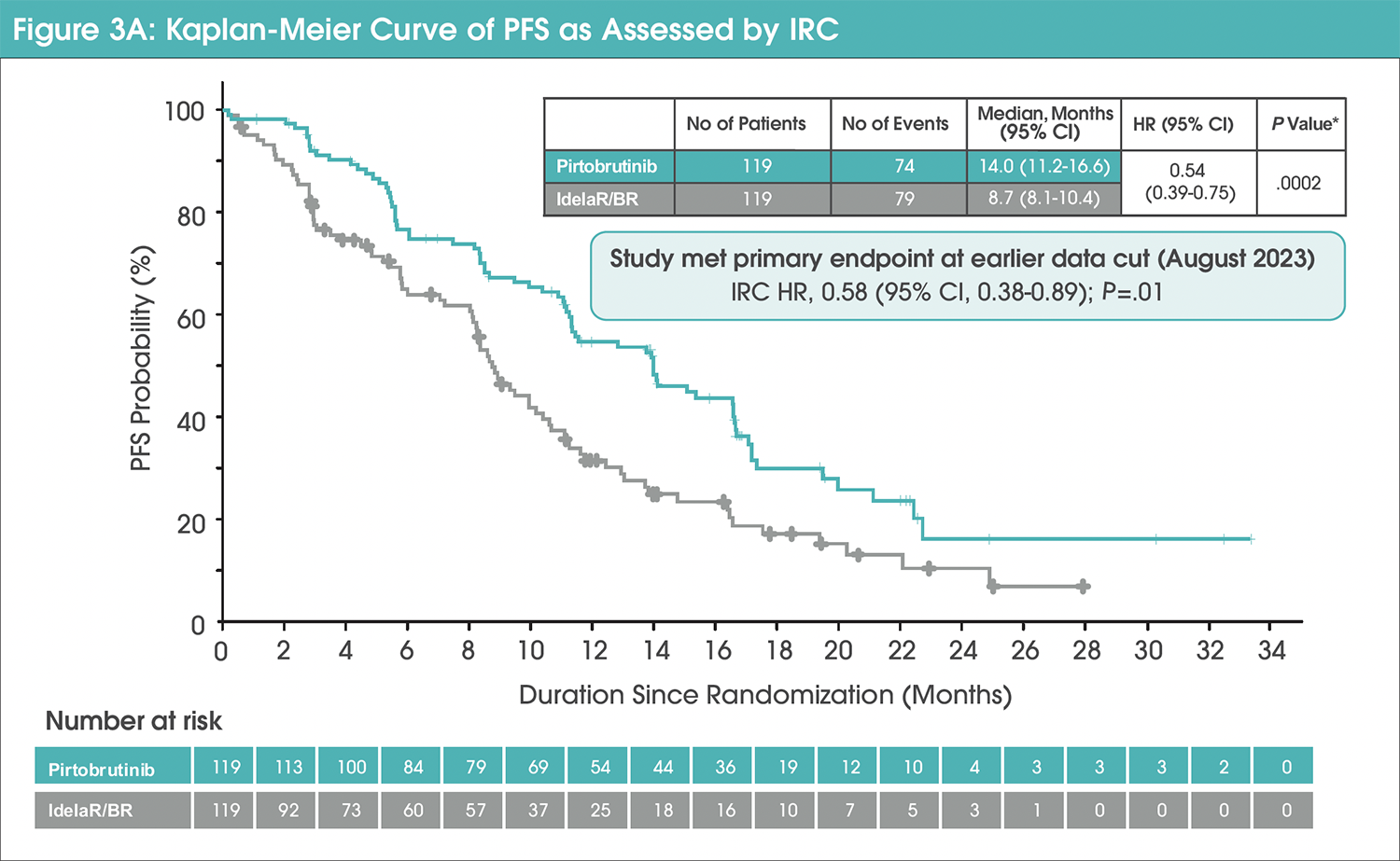
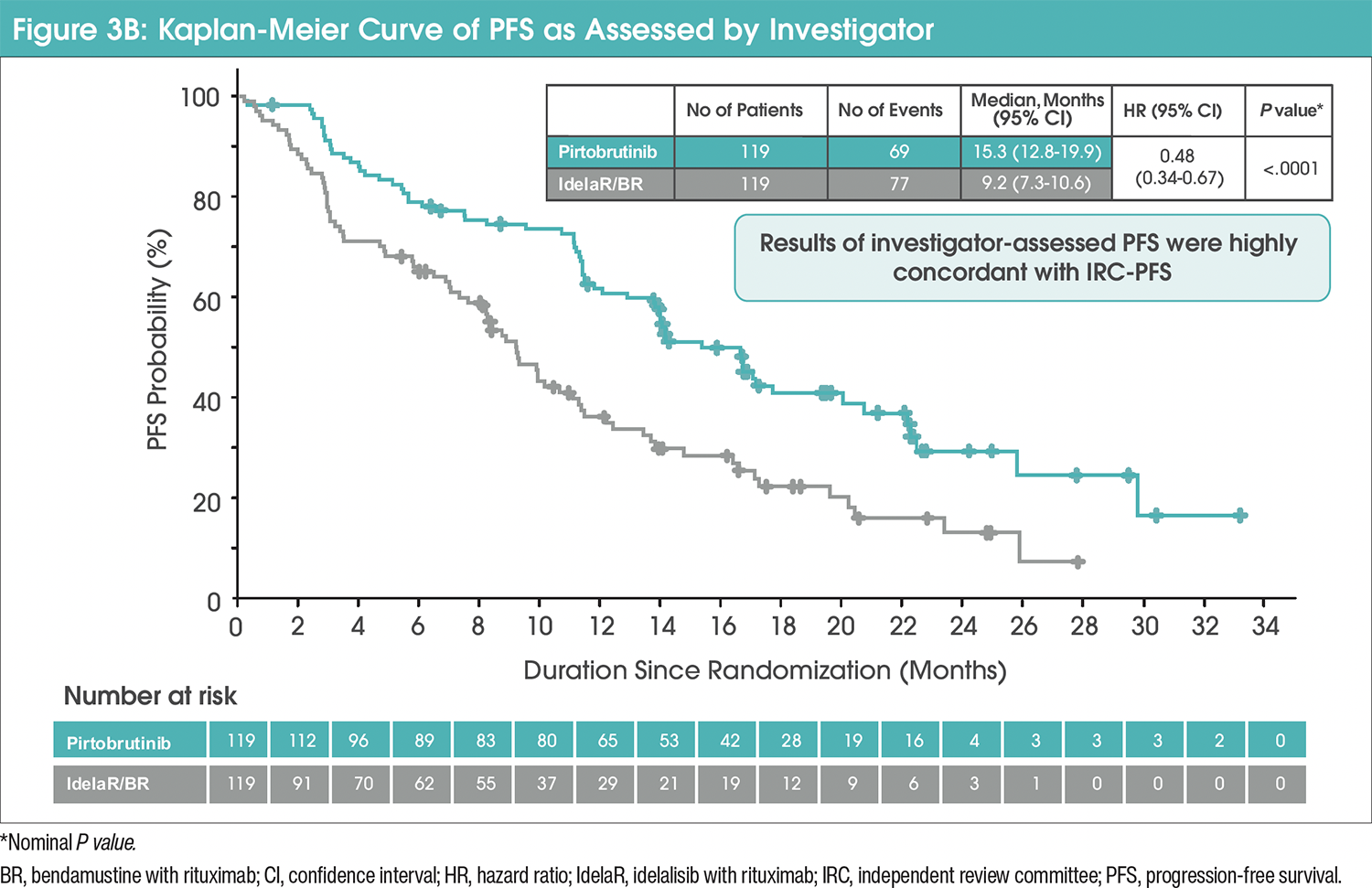
Median IRC-PFS in the pirtobrutinib group was 14 months (95% confidence interval [CI], 11.2-16.6) versus 8.7 months (95% CI, 8.1-10.4) with IdelaR/BR (hazard ratio [HR], 0.54 [95% CI, 0.39-0.75]; nominal P=.0002; Figure 3A).4 The median investigator-assessed PFS was 15.3 months (95% CI, 12.8-19.9) in the pirtobrutinib group and 9.2 months (95% CI, 7.3-10.6) in the IdelaR/BR group (HR, 0.48 [95% CI, 0.34-0.67]; Figure 3B).4
Commentary by Jackie Broadway-Duren, PhD, DNP, APRN, FNP-BC, FAANP: We definitely consider the reason for discontinuation of prior therapy when thinking about subsequent treatment options. For example, if a patient has been on one of the older BTK inhibitors, and we know that the patient is losing that response, then we know that it is likely time to look at another treatment option. Another reason for discontinuation would be if the patient actually starts to develop significant toxicities to the therapy; then, other treatment options are considered at that time.
Commentary by Mollie Moran, APRN-CNP: When considering subsequent treatment options we need to think about why the previous therapy was discontinued. If it's for something like intolerance, is that same potential intolerance going to be there in the next therapeutic option?
To manage patients who are intolerant to covalent BTKis, it is important to consider what their intolerance is. If a patient is on a BTKi and they complain of headaches, and all of their workup is negative for any other cause, then this is due to the BTKi. You will then need to consider if they will have headaches while on subsequent BTKis. It needs to be considered if the category of medication needs to be changed altogether.
Patients’ most common treatment goals in the setting of post–covalent BTKi CLL are to stay in remission and not need further treatment. That is always their goal and ours also. In the event that patients do require additional treatment, who do progress, the goal is to give them the least debilitating treatments. Our goal is to try to keep the patient in remission as long as possible and when they’ve been in remission for a couple of years, our next goal is to try to transition those patients to survivorship where they are still monitored.
We try to always speak with our patients to see what their preferences are. Sometimes it’s not always feasible what their preferences are, but we try to offer them. From a clinician’s standpoint, we want PFS data, as well as time to any new treatment. The most important endpoint would be PFS, which is what we hope for with all the patients, and this is what their desires are as well.
The median TTNT was longer in the pirtobrutinib group at 24 months (95% CI, 17.8-29.7) versus 10.9 months (95% CI, 8.7-12.5) with IdelaR/BR (HR, 0.37 [95% CI, 0.25-0.52]).4 In venetoclax-naïve patients, the median TTNT was longer in the pirtobrutinib group, with a median TTNT of 29.5 months (95% CI, 18.2-not estimable [NE]) versus 12.5 months (95% CI, 9.5-18.4) with IdelaR/BR (HR, 0.36 [95% CI, 0.21-0.61]). In venetoclax-treated patients, there was a longer median TTNT with 20 months (95% CI, 12-NE) in the pirtobrutinib group versus 8.7 months (95% CI, 4.8-11.1) with IdelaR/BR (HR, 0.37 [95% CI, 0.23-0.60]).4
Commentary by Mollie Moran, APRN-CNP: A patient’s most common treatment goals in the setting of post–covalent BTKi CLL is extended TTNT, to not require treatment for a long time. TTNT depends on where you are in the lines of therapy. If you do not need another treatment for a year and a half or 2 years, that would be TTNT, and that could be a significant amount of time before you need to change to another therapy. If you’re in the third-line, fourth-line, or fifth-line therapies, then TTNT tends to get shorter, and so you need to be able to move on to the next therapy without a washout period and without a risk of more significant toxicities.
At the prespecified final OS analysis, 18-month OS was 73.4% (95% CI, 63.9-80.7) in the pirtobrutinib group and 70.8% (95% CI, 60.9-78.7) with IdelaR/BR (unadjusted HR, 1.09 [95% CI, 0.68-1.75]; P=.7202).4 The OS assessment was confounded by a high effective crossover rate of 76%.
Commentary by Mollie Moran, APRN-CNP: OS is important when we look at next therapies, but also time to progression and TTNT. There could be progression, but patients might not require treatment for quite a bit. The distance between those points can be significant. If we can get control over a patient’s disease without having to change therapies, that may be as significant as a PFS endpoint. Response and time to progression and TTNT are important when we are using therapies, especially in the second, third line, and even fourth line in a patient, because you may be using those as bridges.
Treatment-emergent adverse events (TEAEs) of any grade occurred in 108 (93.1%) patients receiving pirtobrutinib and 107 (98.2%) receiving IdelaR/BR.4 Pneumonia was the most frequently occurring TEAE in patients receiving pirtobrutinib (22.4%) versus IdelaR/BR (11.9%), followed by anemia (19.8%) and neutropenia (18.1%). The most frequently occurring adverse events (AEs) in patients receiving IdelaR/BR were diarrhea (31.2%), pyrexia (26.6%), and fatigue and nausea (20.2% each). To account for differences in median treatment durations—15.1 months for pirtobrutinib versus shorter durations with IdelaR (idelalisib, 7.1 months; rituximab, 5.5 months) and BR (bendamustine, 4.7 months; rituximab, 4.7 months)—an exposure-adjusted safety analysis was performed. Treatment discontinuation due to an AE occurred in 20 (17.2%) patients receiving pirtobrutinib, with 6 (5.2%) due to an AE considered treatment-related. Thirty-eight (34.9%) patients receiving IdelaR/BR discontinued treatment due to AEs, with 23 (21.1%) considered treatment-related.4
Commentary by Jackie Broadway-Duren, PhD, DNP, APRN, FNP-BC, FAANP: In this patient population that is treated with pirtobrutinib, we do not see a lot of AEs. The most common AEs that we are challenged with in patients who are on this therapy would be anemia and neutropenia. Cardiac toxicity is one of the reasons why many people are no longer on ibrutinib.
In the literature, pneumonia was shown to be one of the most likely AEs, occurring in about 20% of patients, but we do not see a lot of pneumonia in our patient population. However, pneumonia may be more typical in patients who have comorbidities that are already at risk for that. To mitigate pneumonia, we try to get these patients up-to-date on their pneumonia vaccines.
Commentary by Mollie Moran, APRN-CNP: The safety profile of pirtobrutinib is well established. From the BRUIN trial and other trials, the common side effects include infection, risk of infection, risk of bleeding, and cytopenias. For those who treat CLL, these are complications that are seen when starting any sort of CLL therapy, especially when a patient is progressing. The risk of atrial fibrillation seems to be slightly less in the pirtobrutinib population. We keep an eye on their liver function when they first start pirtobrutinib. There can be some muscle pain too when changing therapies; they tend to produce myalgias and joint aches, so that is manageable and well known. As initial therapy, there tends to be some [gastrointestinal] toxicity, so we manage diarrhea and nausea with either prescription medications or over-the-counter medications. It is important to get a baseline assessment of a patient before starting them on one of these therapies.
Conclusion
The BRUIN-321 showed the benefit of pirtobrutinib in patients previously treated with a covalent BTKi. This study showed that pirtobrutinib provided a significant and clinically meaningful improvement in PFS along with a more favorable safety profile compared with IdelaR/BR in patients with CLL/SLL.
Considerations for clinical practice
Currently, no prospective randomized controlled studies have been conducted exclusively in patients who have been previously treated with a covalent BTKi. The ASCEND study evaluated acalabrutinib in BTK-naïve R/R patients with CLL/SLL compared with the same control group of IdelaR/BR.18 The phase 3 MURANO study compared venetoclax (a BCL2i) in combination with rituximab with a standard chemoimmunotherapy in patients with R/R CLL.19 Very few patients, 2.6%, were previously treated with a BTKi. Following disease progression, 3 patients in the venetoclax-rituximab group and 40 patients in the BR group received targeted therapies for CLL, including B-cell receptor signaling inhibitors and BCL2is.19 The BRUIN CLL-321 study, however, evaluated patients with R/R CLL/SLL previously treated with, but not limited to, covalent BTKis, chemotherapy, and immunotherapy.4 The risk profile of post-BTKi patients may differ from that of BTKinaïve patients, as post-BTKi patients may have more del(17p) and/or TP53 mutations, complex karyotype, and IGHV unmutated.4,18
TTNT is measured as the duration from the initiation of a specific therapy to the start of the subsequent line of treatment.20 It serves as a real-world clinical endpoint to assess treatment durability, capturing both the therapy’s effectiveness and tolerability.20 PFS is measured as the time from the start of treatment or randomization in a clinical trial to either the first documented disease progression or death from any cause, whichever occurs first.21
PFS in CLL can be significantly influenced by the method and frequency of disease monitoring used to monitor disease progression, whether through clinical exams alone or routine radiologic assessments. In clinical trials, progression is typically monitored using a combination of clinical exams, laboratory tests, and imaging studies to ensure comprehensive and accurate assessments.22
The iwCLL guidelines provide clear criteria for initiating therapy and managing progression based on clinical and laboratory findings. Progression is categorized as either asymptomatic or clinically meaningful, and treatment decisions are tailored accordingly. Patients with asymptomatic progression are typically managed with observation rather than immediate intervention.22 iwCLL guidelines recommend against starting therapy in the absence of symptoms or complications, as early intervention has not shown a survival benefit in asymptomatic patients.22 Clinically meaningful progression is characterized by symptoms or complications that impact quality of life or organ function. iwCLL guidelines recommend starting treatment when patients meet specific criteria for clinically meaningful progression, such as symptomatic disease or organ dysfunction.22 First-line treatment typically involves BTKis or BCL2 is based on patient-specific factors and preferences.3
Commentary by Jackie Broadway-Duren, PhD, DNP, APRN, FNP-BC, FAANP: We follow NCCN guidelines, as well as consider the patient individually to determine what treatment might be best for that patient. We also consider guidelines from the iwCLL as well to guide treatment decisions.
Commentary by Mollie Moran, APRN-CNP: Treatment guidelines are helpful when deciding when to start treatment, and the sequencing is certainly important when looking at who has high-risk disease versus who doesn't have high-risk disease based on mutational status, and whether or not somebody has a 17p or a p53 mutation. Guidelines are helpful in that regard.
Shared decision-making is a collaborative process where patients and clinicians work together to choose treatments that align with the patient's clinical needs, lifestyle priorities, and personal preferences.23 In the context of CLL, shared decision-making often involves weighing the trade-offs between fixed-duration therapies and continuous treatments, as well as the differences in treatment modalities, such as intravenous (IV) versus oral. Key considerations include the mechanism of action, treatment burden, monitoring requirements, and patient-specific factors.24 Fixed-duration regimens (eg, venetoclax combined with anti-CD20 monoclonal antibodies like rituximab or obinutuzumab) typically involve a defined treatment period, often 12 to 24 months.19 Continuous treatments, such as oral covalent BTKis like ibrutinib, acalabrutinib, or zanubrutinib, and noncovalent BTKis like pirtobrutinib, involve indefinite daily administration until disease progression or unacceptable toxicity.4,25 Most oral therapies typically require fewer clinic visits, with monitoring conducted through periodic lab tests (eg, monthly or biannually). This approach minimizes disruption to a patient’s daily routine. Some oral therapies, however, are given for a fixed duration but require increased clinic visits and close monitoring due to the risk of tumor lysis syndrome.26 Oral medications are self-administered, offering convenience and flexibility for patients who prioritize independence.27 IV therapies require regular in-clinic visits for administration, often weekly or biweekly during the initial phase. This increases the time burden for patients and caregivers. IV administration is managed by healthcare professionals, which can be reassuring, but also logistically challenging for patients with mobility or transportation issues.
Commentary by Mollie Moran, APRN-CNP: We approach follow-up monitoring by continuing to do frequent labs or clinic visits. If someone is progressing standardly, we would do 3-month visits, unless their progression is much quicker; then, we would do monthly visits or in-between lab visits to make sure that their disease isn’t progressing. We watch white blood cell counts to make sure it is not doubling, that hemoglobin isn’t dropping below 8, and that the platelets aren’t dropping below 100. The time period in between those visits varies based on the projection of those numbers. We may see people more frequently as they are progressing on current therapy in a standard follow-up of every 6 months or once a year.
Treatment sequencing for CLL and SLL is a critical aspect of disease management, as patients often require multiple lines of therapy over the course of their disease. Optimal sequencing depends on several factors, including disease biology, prior treatments, treatment responses, comorbidities, and patient preferences. Clinical guidelines recommend targeted therapies as the preferred first-line options for most patients with CLL/SLL, and preferred regimens include a covalent BTKi or BCL2i.3 Preferred second-line options include venetoclax-based therapy and covalent and noncovalent BTKis.3
The management of CLL/SLL with a noncovalent BTKi remains a nuanced and evolving area of clinical practice, guided by available evidence and expert recommendations. Real-world endpoints such as TTNT and PFS further support the evaluation of therapeutic durability and effectiveness. The iwCLL guidelines serve as an essential framework for treatment initiation and disease monitoring, emphasizing the importance of tailoring interventions based on clinical progression and patient-specific factors. Close monitoring and regular follow-up visits allow clinicians to make individualized treatment plans for patients upon disease progression. Shared decision-making remains central to optimizing outcomes, ensuring that treatment choices align with the patient’s clinical needs, quality-of-life considerations, and personal preferences. As new therapies and evidence continue to emerge, clinicians must remain adaptable, leveraging both clinical guidelines and individualized care approaches to address the complex needs of patients with CLL/SLL.
Commentary by Jackie Broadway-Duren, PhD, DNP, APRN, FNP-BC, FAANP: I think the addition of pirtobrutinib has opened the door for better outcomes in our patients. Before this drug was FDA approved, we thought it would be a disadvantage for the patient if they progress on a covalent BTKi and could no longer target the BTKi pathway. So, we're happy to see pirtobrutinib come along for the treatment of covalent BTKi–pretreated CLL/SLL.
References
- National Cancer Institute. The Surveillance, Epidemiology, and End Results (SEER) Program. Cancer Stat Facts: Leukemia—Chronic Lymphocytic Leukemia (CLL). 2024. Accessed October 29, 2025. https://seer.cancer.gov/statfacts/html/clyl.html
- National Cancer Institute. CLL/SLL. Accessed August 4, 2025. www.cancer.gov/publications/dictionaries/cancer-terms/def/cll-sll
- Wierda WG, Brown J, Abramson JS, et al. NCCN Guidelines Insights: Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma, Version 3.2022. J Natl Compr Canc Netw. 2022;20:622-634.
- Sharman JP, Munir T, Grosicki S, et al. Phase III trial of pirtobrutinib versus idelalisib/rituximab or bendamustine/rituximab in covalent Bruton tyrosine kinase inhibitor-pretreated chronic lymphocytic leukemia/small lymphocytic lymphoma (BRUIN CLL-321). J Clin Oncol. 2025;43:2538-2549.
- Smith TW, Owusu HF, Wormser D, et al. Real-world evaluation of the treatment landscape for chronic lymphocytic leukemia. Blood. 2021;138:1559.
- Shadman M. Diagnosis and treatment of chronic lymphocytic leukemia: a review. JAMA. 2023;329:918-932.
- Mato AR, Woyach JA, Brown JR, et al. Pirtobrutinib after a covalent BTK inhibitor in chronic lymphocytic leukemia. N Engl J Med. 2023;389:33-44.
- Woyach JA, Brown JR, Ghia P, et al. Pirtobrutinib in post-cBTKi CLL/SLL: ~30 months follow-up and subgroup analysis with/without prior BCL2i from the phase 1/2 BRUIN study. Blood. 2023;142(Suppl 1):325-325.
- US Food & Drug Administration. FDA grants accelerated approval to pirtobrutinib for chronic lymphocytic leukemia and small lymphocytic lymphoma. 2023. Accessed November 4, 2024. www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-pirtobrutinib-chroniclymphocytic- leukemia-and-small-lymphocytic
- Hendriks RW, Yuvaraj S, Kil LP. Targeting Bruton's tyrosine kinase in B cell malignancies. Nat Rev Cancer. 2014;14:219-232.
- Gu D, Tang H, Wu J, et al. Targeting Bruton tyrosine kinase using non-covalent inhibitors in B cell malignancies. J Hematol Oncol. 2021;14:40.
- Brullo C, Villa C, Tasso B, et al. Btk inhibitors: a medicinal chemistry and drug delivery perspective. Int J Mol Sci. 2021;22:7641.
- Tambaro FP, De Novellis D, Wierda WG. The role of BTK inhibition in the treatment of chronic lymphocytic leukemia: a clinical view. J Exp Pharmacol. 2021;13:923-935.
- Aljoundi A, Bjij I, El Rashedy A, et al. Covalent versus non-covalent enzyme inhibition: which route should we take? A justification of the good and bad from molecular modelling perspective. Protein J. 2020;39:97-105.
- Gomez EB, Ebata K, Randeria HS, et al. Preclinical characterization of pirtobrutinib, a highly selective, noncovalent (reversible) BTK inhibitor. Blood. 2023;142:62-72.
- Lewis KL, Cheah CY. Non-covalent BTK inhibitors-the new BTKids on the block for B-cell malignancies. J Pers Med. 2021;11:764.
- Tasso B, Spallarossa A, Russo E, et al. The development of BTK inhibitors: a five-year update. Molecules. 2021;26:7411.
- Ghia P, Pluta A, Wach M, et al. ASCEND: phase III, randomized trial of acalabrutinib versus idelalisib plus rituximab or bendamustine plus rituximab in relapsed or refractory chronic lymphocytic leukemia. J Clin Oncol. 2020;38:2849-2861.
- Seymour JF, Kipps TJ, Eichhorst B, et al. Venetoclax–rituximab in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2018;378:1107-1120.
- Campbell BA, Scarisbrick JJ, Kim YH, et al. Time to next treatment as a meaningful endpoint for trials of primary cutaneous lymphoma. Cancers. 2020;12:2311.
- Gutman SI, Piper M, Grant MD, et al. Progression-free survival: what does it mean for psychological well-being or quality of life? [Internet]. Agency for Healthcare Research and Quality (US). 2013. Accessed October 29, 2025. www.ncbi.nlm.nih.gov/books/NBK137763/
- Hallek M, Cheson BD, Catovsky D, et al. iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood. 2018;131:2745-2760.
- Montori VM, Ruissen MM, Hargraves IG, et al. Shared decision-making as a method of care. BMJ Evid Based Med. 2023;28:213-217.
- Eichhorst B, Robak T, Montserrat E, et al. Chronic lymphocytic leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2021;32:23-33.
- Montoya S, Thompson MC. Non-covalent Bruton's tyrosine kinase inhibitors in the treatment of chronic lymphocytic leukemia. Cancers (Basel). 2023;15:3648.
- Gribben JG. Practical management of tumour lysis syndrome in venetoclax-treated patients with chronic lymphocytic leukaemia. Br J Haematol. 2020;188:844-851.
- Fifer S, Godsell J, Opat S, et al. Understanding the experience, treatment preferences and goals of people living with chronic lymphocytic leukemia (CLL) in Australia. BMC Cancer. 2024;24:831.